dsp.BlockLMSFilter
Compute output, error, and weights using block least mean squares (LMS) adaptive algorithm
Description
The dsp.BlockLMSFilter
System object™ computes output, error, and weights using the block LMS adaptive
algorithm.
To compute the output, error, and weights:
Create the
dsp.BlockLMSFilterobject and set its properties.Call the object with arguments, as if it were a function.
To learn more about how System objects work, see What Are System Objects?
Creation
Syntax
Description
blms = dsp.BlockLMSFilterblms, that filters the input signal and computes
filter weights based on the block least mean squares (LMS) algorithm.
blms = dsp.BlockLMSFilter(length,blocksize)blms, with the
Length property set to length and the
BlockSize property set to blocksize.
blms = dsp.BlockLMSFilter(PropertyName=Value)BlockSize to 16.
Properties
Usage
Syntax
Description
[
filters input y,err,wts] = blms(x,d,mu,a,r)x, using d as the desired signal,
mu as the step size, a as the adaptation
control, and r as the reset signal. The object returns the filtered
output y, the filter error err, and the adapted
filter weights wts. Set the properties appropriately to provide all
possible inputs.
Input Arguments
Output Arguments
Object Functions
To use an object function, specify the
System object as the first input argument. For
example, to release system resources of a System object named obj, use
this syntax:
release(obj)
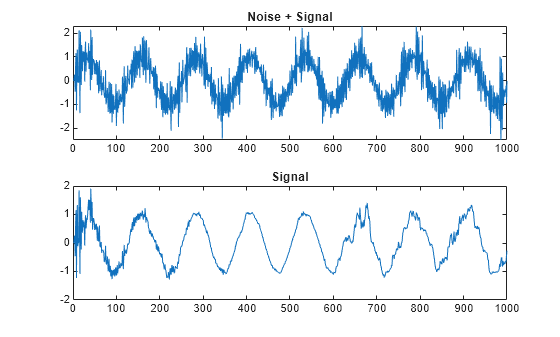
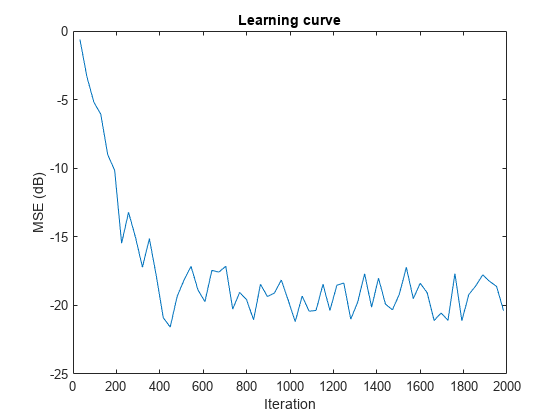
Examples
Algorithms
This object implements the algorithm, inputs, and outputs described on the Block LMS Filter block reference page. The object properties correspond to the block parameters.
Extended Capabilities
Version History
Introduced in R2012a