dsp.AllpassFilter
Single section or cascaded allpass filter
Description
The dsp.AllpassFilter object filters each channel of the input using
allpass filter implementations. To import this object into Simulink®, use the MATLAB® System block.
To filter each channel of the input:
Create the
dsp.AllpassFilterobject and set its properties.Call the object with arguments, as if it were a function.
To learn more about how System objects work, see What Are System Objects?
Creation
Description
Allpass = dsp.AllpassFilterAllpass, that filters each channel of the input signal
independently using an allpass filter, with the default structure and coefficients.
Allpass = dsp.AllpassFilter(PropertyName=Value)Allpass, with each property set to the specified value
by one or more Name-Value pair arguments. Name
is the property name and Value is the corresponding value. For
example, to set the filter structure as "Lattice", set
Structure to "Lattice".
Properties
Unless otherwise indicated, properties are nontunable, which means you cannot change their
values after calling the object. Objects lock when you call them, and the
release function unlocks them.
If a property is tunable, you can change its value at any time.
For more information on changing property values, see System Design in MATLAB Using System Objects.
Internal allpass filter implementation structure, specified as one of these:
"Minimum multiplier""Lattice""Wave Digital Filter"
Each structure uses a different set of coefficients, independently stored in the corresponding object property.
Real allpass polynomial filter coefficients, specified as one of these:
N-by-1 matrix –– N first-order allpass sections.
N-by-2 matrix –– N second-order allpass sections.
N-by-4 matrix –– N fourth-order allpass sections. (since R2024a)
The default value of [-2^(-1/2) 0.5] defines a stable
second-order allpass filter with poles and zeros located at ±π/3 in the
z-plane.
Tunable: Yes
Dependencies
To enable this property, set the
Structure property to "Minimum
multiplier".
Data Types: single | double | int8 | int16 | int32 | int64 | uint8 | uint16 | uint32 | uint64
Real allpass coefficients in the Wave Digital Filter form,
specified as an N-by-1 or
N-by-2 matrix of
N first-order or second-order allpass sections.
All elements must have absolute values less than or equal to
1. This value is a transformed version of the
default value of AllpassCoefficients, computed
using allpass2wdf(AllpassCoefficients). These
coefficients define the same stable second-order allpass filter as
when Structure is set to "Minimum
multiplier".
Tunable: Yes
Dependencies
To enable this property, set the
Structure property to "Wave
Digital Filter".
Data Types: single | double | int8 | int16 | int32 | int64 | uint8 | uint16 | uint32 | uint64
Real or complex allpass coefficients as lattice reflection
coefficients, specified as a row vector (single-section
configuration) or a column vector. This value is a transformed and
transposed version of the default value of
AllpassCoefficients, computed using
transpose(tf2latc([1 h.AllpassCoefficients])).
These coefficients define the same stable second-order allpass
filter as when Structure is set to
"Lattice".
Tunable: Yes
Dependencies
To enable this property, set the
Structure property to
"Lattice".
Data Types: single | double | int8 | int16 | int32 | int64 | uint8 | uint16 | uint32 | uint64
Complex Number Support: Yes
Indicate if last section is first order. When you set the
TrailingFirstOrderSection property to true,
the last section is considered to be first-order, and the second order term in the last
row of the N-by-2 matrix is ignored. In case of the minimum
multiplier structure, if you specify an N-by-4 matrix, this property
has no effect on the filter coefficients.
Dependencies
To enable this property, set the
Structure property to "Minimum
multiplier" or "Wave Digital
Filter".
Usage
Syntax
Description
Input Arguments
Data input, specified as a vector or a matrix. This object also accepts variable-size inputs. Once the object is locked, you can change the size of each input channel, but you cannot change the number of channels.
Data Types: single | double
Complex Number Support: Yes
Output Arguments
Filtered output, returned as a vector or a matrix. The size, data type, and complexity of the output signal matches that of the input signal.
Data Types: double | single
Complex Number Support: Yes
Object Functions
To use an object function, specify the
System object as the first input argument. For
example, to release system resources of a System object named obj, use
this syntax:
release(obj)
freqz | Frequency response of discrete-time filter System object |
filterAnalyzer | Analyze filters with Filter Analyzer app |
impz | Impulse response of discrete-time filter System object |
info | Information about filter System object |
coeffs | Returns the filter System object coefficients in a structure |
cost | Estimate cost of implementing filter System object |
grpdelay | Group delay response of discrete-time filter System object |
ctf | Convert digital filter to coefficients in cascaded transfer function format |
outputDelay | Determine output delay of single-rate or multirate filter |
setInputSampleRate | Specify input sample rate in filter objects |
Examples
Construct the two dsp.AllpassFilter objects.
Fs = 48000; % in Hz FL = 1024; APF1 = dsp.AllpassFilter(AllpassCoefficients=[-0.710525516540603 0.208818210000029]); APF2 = dsp.AllpassFilter(AllpassCoefficients=[-0.940456403667957 0.6;... -0.324919696232907 0],... TrailingFirstOrderSection=true);
Construct the Transfer Function Estimator to estimate the transfer function between the random input and the Allpass filtered output.
TFE = dsp.TransferFunctionEstimator(FrequencyRange="onesided",... SpectralAverages=2);
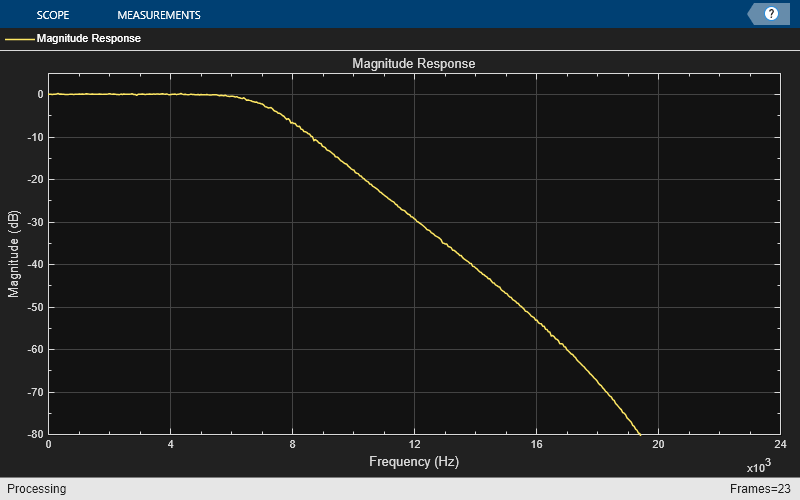
Construct the dsp.ArrayPlot object to plot the magnitude response.
AP = dsp.ArrayPlot(PlotType="Line",YLimits=[-80 5],... YLabel="Magnitude (dB)",SampleIncrement=Fs/FL,... XLabel="Frequency (Hz)",Title="Magnitude Response",... ShowLegend=true,ChannelNames={"Magnitude Response"});
Filter the Input and show the magnitude response of the estimated transfer function between the input and the filtered output.
tic; while toc < 5 in = randn(FL,1); out = 0.5.*(APF1(in) + APF2(in)); A = TFE(in, out); AP(db(A)); end
Since R2024a
Design a quasi-linear IIR halfband filter with the order of 32 using the designHalfbandIIR function. Assign the filter coefficients to a coupled allpass filter.
[a0,a1] = designHalfbandIIR(FilterOrder=32,DesignMethod="quasilinphase",Verbose=true)designHalfbandIIR(FilterOrder=32, TransitionWidth=0.1, DesignMethod="quasilinphase", Structure="single-rate", InputSampleRate="normalized", Datatype="double", SystemObject=false)
a0 = 4×4
0 0 0 0
0 0 0 0
0 0 0 0
0 0 0 0
a1 = 4×4
0 0.8085 0 0.3051
0 0.0387 0 0.2695
0 -0.7054 0 0.2604
0 0.3546 0 -0.4386
Construct the corresponding coupled allpass filter using the cascade and parallel functions. Alternatively, if you set the SystemObject argument of the designHalfbandIIR function to true, the function designs the same object.
B0 = dsp.AllpassFilter(AllpassCoefficients=a0); B1 = dsp.AllpassFilter(AllpassCoefficients=a1); filtObj = cascade(parallel(B0,cascade(dsp.Delay, B1)),0.5)
filtObj =
dsp.FilterCascade with properties:
Stage1: [1×1 dsp.ParallelFilter]
Stage2: 0.5000
CloneStages: true
Create a dsp.DynamicFilterVisualizer object and visualize the magnitude response of the filter.
dfv = dsp.DynamicFilterVisualizer(NormalizedFrequency=true); dfv(filtObj);

A quasi-linear IIR filter has a fairly constant group delay (hence almost linear phase) in the passband region of the filter.
grpdelay(filtObj)

phasez(filtObj)

Create a spectrumAnalyzer object to visualize the spectra of the input and output signals.
scope = spectrumAnalyzer(SampleRate=2, ... PlotAsTwoSidedSpectrum=false,... ChannelNames=["Input Signal","Filtered Signal"]);
Stream in a noisy sinusoidal signal and filter the signal using the IIR halfband filter. The sinusoidal tone falls in the passband frequency of the filter and is therefore unaffected.
sine = dsp.SineWave(Frequency=1000,SampleRate=44100,SamplesPerFrame=1024)
sine =
dsp.SineWave with properties:
Amplitude: 1
Frequency: 1000
PhaseOffset: 0
ComplexOutput: false
Method: 'Trigonometric function'
SamplesPerFrame: 1024
SampleRate: 44100
OutputDataType: 'double'
for i = 1:1000 x = sine()+0.005*randn(1024,1); y = filtObj(x); scope(x,y); end

Design a Butterworth IIR halfband filter with the order of 13 and construct the corresponding coupled allpass filter using the designHalfbandIIR function. Specify the input sample rate as 1200 Hz. Set the SystemObject argument of the function to true.
Fs = 1200;
filtObj = designHalfbandIIR(FilterOrder=13,InputSampleRate=Fs,...
Verbose=true,SystemObject=true)designHalfbandIIR(FilterOrder=13, DesignMethod="butter", Structure="single-rate", InputSampleRate=1200, Datatype="double", SystemObject=true, Passband="lowpass")
filtObj =
dsp.FilterCascade with properties:
Stage1: [1×1 dsp.ParallelFilter]
Stage2: 0.5000
CloneStages: true
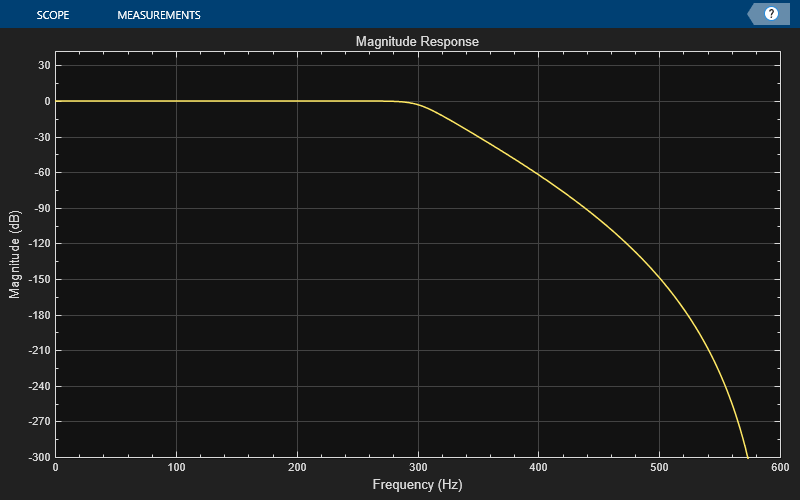
Create a dsp.DynamicFilterVisualizer object and visualize the magnitude response of the filter.
dfv = dsp.DynamicFilterVisualizer(SampleRate=Fs); dfv(filtObj);
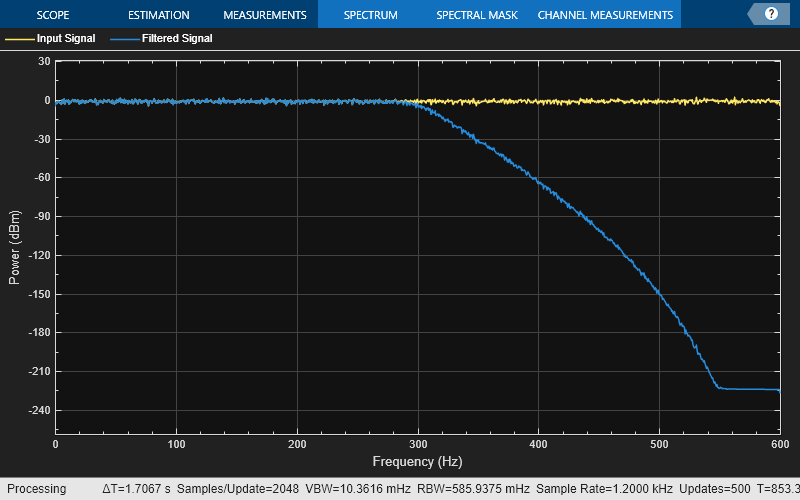
Create a spectrumAnalyzer object to visualize the spectra of the input and output signals.
scope = spectrumAnalyzer(SampleRate=Fs, ... PlotAsTwoSidedSpectrum=false,... ChannelNames=["Input Signal","Filtered Signal"]);
Stream in random data and filter the signal using the IIR halfband filter.
for i = 1:1000 x = randn(1024, 1); y = filtObj(x); scope(x,y); end
Since R2026a
Create a dsp.AllpassFilter object and specify the allpass polynomial coefficients.
apFilt = dsp.AllpassFilter(AllpassCoefficients=[0 0.5539 0 0.2503;
0 -0.4364 0 0.2218;
0 0.3781 0 -0.3933])apFilt =
dsp.AllpassFilter with properties:
Structure: 'Minimum multiplier'
AllpassCoefficients: [3×4 double]
TrailingFirstOrderSection: false
Use the ctf function to obtain coefficients in the CTF format and the scale values.
[apnum,apden,sv] = ctf(apFilt)
apnum = 3×5
0.2503 0 0.5539 0 1.0000
0.2218 0 -0.4364 0 1.0000
-0.3933 0 0.3781 0 1.0000
apden = 3×5
1.0000 0 0.5539 0 0.2503
1.0000 0 -0.4364 0 0.2218
1.0000 0 0.3781 0 -0.3933
sv = 1
Algorithms
The transfer function of an allpass filter is given by
.
c is allpass polynomial coefficients vector. The order, n, of the transfer function is the length of vector c.
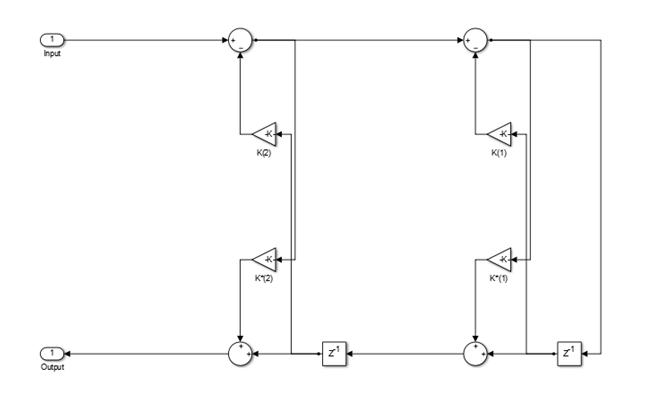
In the minimum multiplier form and wave digital form, the allpass filter is implemented as a cascade of either second-order (biquad) sections or first-order sections. When the coefficients are specified as an N-by-2 matrix, each row of the matrix specifies the coefficients of a second-order filter. The last element of the last row can be ignored based on the trailing first-order setting. When the coefficients are specified as an N-by-1 matrix, each element in the matrix specifies the coefficient of a first-order filter. The cascade of all the filter sections forms the allpass filter.
In the lattice form, the coefficients are specified as a vector.
These structures are computationally more economical and structurally more stable compared to the generic IIR filters, such as df1, df1t, df2, df2t. For all structures, the allpass filter can be a single-section or a multiple-section (cascaded) filter. The different sections can have different orders, but they are all implemented according to the same structure.
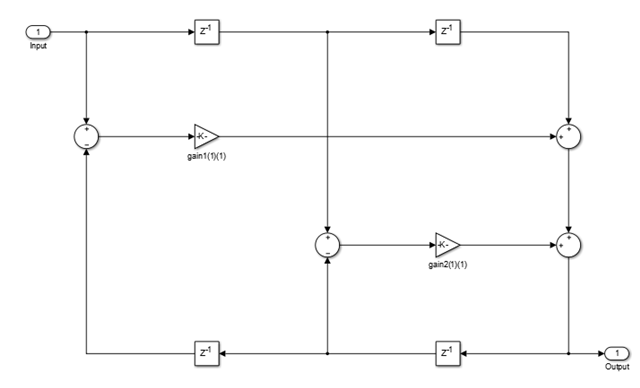
This structure realizes the allpass filter with the minimum number of required multipliers,
equal to the order n. It also uses 2n delay units
and 2n adders. The multipliers uses the specified coefficients, which
are equal to the polynomial vector c in the allpass transfer
function.
In this second-order section of the minimum multiplier structure, the coefficients
vector, c, is equal to [0.1 -0.7].
In this fourth-order section of the minimum multiplier structure, the coefficients
vector, c, is equal to [-0.7071 0.5 0.4
0.25].
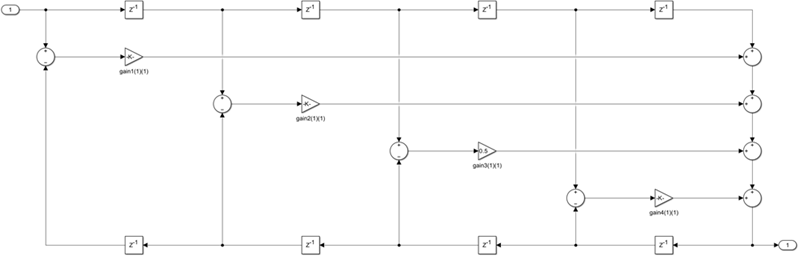
This structure uses n multipliers, but only n delay
units, at the expense of requiring 3n adders. To
use this structure, specify the coefficients in wave digital filter
(WDF) form. Obtain the WDF equivalent of the conventional allpass
coefficients using allpass2wdf(allpass_coefficients).
To convert WDF coefficients into the equivalent allpass polynomial
form, use wdf2allpass(WDF coefficients). In this
second-order section of the WDF structure, the coefficients vector w is
equal to allpass2wdf([0.1 -0.7]).
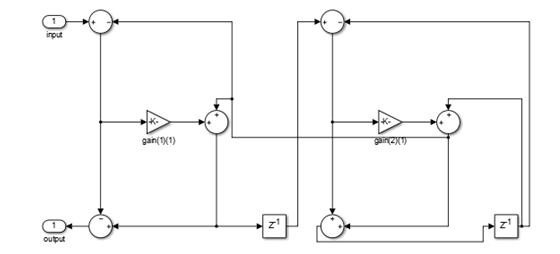
This lattice structure uses 2n multipliers, n delay
units, and 2n adders. To use this structure, specify
the coefficients as a vector.
You can obtain the lattice equivalent of the conventional allpass
coefficients using transpose(tf2latc(1, [1 allpass_coefficients])).
In the following second-order section of the lattice structure, the
coefficients vector is computed using transpose(tf2latc(1,
[1 0.1 -0.7])). Use these coefficients for a filter that
is functionally equivalent to the minimum multiplier structure with
coefficients [0.1 -0.7].
References
[1] Regalia, Philip A. and Mitra Sanjit K. and Vaidyanathan, P. P. (1988) “The Digital All-Pass Filter: A Versatile Signal Processing Building Block.” Proceedings of the IEEE, Vol. 76, No. 1, 1988, pp. 19–37
[2] M. Lutovac, D. Tosic, B. Evans, Filter Design for Signal Processing Using MATLAB and Mathematica. Upper Saddle River, NJ: Prentice Hall, 2001.
Extended Capabilities
Usage notes and limitations:
The System object supports code generation only when the
Structureproperty is set toMinimum multiplierorLattice.See System Objects in MATLAB Code Generation (MATLAB Coder).
Version History
Introduced in R2013aWhen you set the Structure property to 'Minimum
multipler', you can specify an N-by-4 matrix of filter
coefficients in the AllpassCoefficients property, where
N is the number of filter sections.
See Also
Functions
Objects
MATLAB Command
You clicked a link that corresponds to this MATLAB command:
Run the command by entering it in the MATLAB Command Window. Web browsers do not support MATLAB commands.
Select a Web Site
Choose a web site to get translated content where available and see local events and offers. Based on your location, we recommend that you select: .
You can also select a web site from the following list
How to Get Best Site Performance
Select the China site (in Chinese or English) for best site performance. Other MathWorks country sites are not optimized for visits from your location.
Americas
- América Latina (Español)
- Canada (English)
- United States (English)
Europe
- Belgium (English)
- Denmark (English)
- Deutschland (Deutsch)
- España (Español)
- Finland (English)
- France (Français)
- Ireland (English)
- Italia (Italiano)
- Luxembourg (English)
- Netherlands (English)
- Norway (English)
- Österreich (Deutsch)
- Portugal (English)
- Sweden (English)
- Switzerland
- United Kingdom (English)